- CMS: Medicare Program; Prospective Payment System and Consolidated Billing for Skilled Nursing Facilities; Updates to the Quality Reporting Program for Federal Fiscal Year 2026
- CMS: Medicare Program; FY 2026 Hospice Wage Index and Payment Rate Update and Hospice Quality Reporting Program Requirements
- Public Inspection: CMS: Medicare Program: Prospective Payment System and Consolidated Billing for Skilled Nursing Facilities; Updates to the Quality Reporting Program for Federal Fiscal Year 2026
- Public Inspection: CMS: Medicare Program: Fiscal Year 2026 Hospice Wage Index and Payment Rate Update and Hospice Quality Reporting Program Requirements
- CMS: Request for Information; Health Technology Ecosystem
- VA: Staff Sergeant Fox Suicide Prevention Grant Program Funding Opportunity
- State: 60-Day Notice of Proposed Information Collection: J-1 Visa Waiver Recommendation Application
- Public Inspection: CMS: Request for Information: Health Technology Ecosystem
- HHS: Request for Information (RFI): Ensuring Lawful Regulation and Unleashing Innovation To Make American Healthy Again
- VA: Solicitation of Nominations for the Appointment to the Advisory Committee on Tribal and Indian Affairs
- GAO Seeks New Members for Tribal and Indigenous Advisory Council
- VA: Staff Sergeant Fox Suicide Prevention Grant Program Funding Opportunity
- Telehealth Study Recruiting Veterans Now
- USDA Delivers Immediate Relief to Farmers, Ranchers and Rural Communities Impacted by Recent Disasters
- Submit Nominations for Partnership for Quality Measurement (PQM) Committees
Vaccine Order in Pennsylvania Signed Feb. 12
Last week, Acting Pennsylvania Secretary of Health Beam signed an order outlining appropriate steps and recognized best practices to ensure vaccine providers deliver 80 percent of doses within seven days of receipt, provide a phone number where people can speak to an individual to make an appointment, and report race and ethnicity data for everyone vaccinated. If you have questions about the state’s vaccine distribution or new order, contact Eric Kiehl, PACHC Director of Policy and Partnerships. PACHC has already submitted a series of questions to DOH on the order and Sec. Beam has committed to providing a written response.
Pennsylvania Takes Action to Ensure Pennsylvanians Get Second Doses of COVID-19 Vaccine
The Pennsylvania Department of Health (DOH) and the newly formed joint task force with the legislature this week reaffirmed their commitment to ensuring that Pennsylvanians will have access to second doses of COVID-19 vaccine within the CDC-recommended timeframe of up to 42 days after the first dose. Acknowledging communications shortcomings and the need for more frequent outreach to providers, Acting Secretary of Health Alison Beam outlined plans moving forward. As DOH reviewed and worked to improve the complex processes necessary to get COVID-19 vaccine from the manufacturers into the arms of Pennsylvanians as quickly as possible, they discovered some providers inadvertently administered the Moderna vaccine shipped to them intended as second doses, as first doses. To remedy the situation and remain committed to ensuring that second doses are available, DOH is adjusting the timing of second dose administration following CDC guidelines that set the minimum time between doses at 28 days and the maximum time at 42 days.
How Are We Doing with COVID-19 Vaccination?
Since vaccine distribution began in the U.S. on Dec. 14, more than 55 million doses have been administered, reaching 11.9 percent of the total U.S. population, according to federal data collected by the Centers for Disease Control and Prevention (CDC). The U.S. is currently administering more than 1.8 million shots a day. States will receive 13.5 million doses this week, up from 11 million nationwide last week and a 57 percent increase since President Biden took office. With the recent purchase of 200 million additional doses by the administration, it is projected that the U.S. is on target to have enough supply for 300 million Americans by the end of July. In Pennsylvania, 10.7 percent of the population has received at least one dose and 3.6 percent, two doses. That is assuming that all vaccine administrators have inputted their data in PA-SIIS, which is not the case and is one of the reasons the state is reconfiguring its distribution and vaccination strategy. Over the course of the next few weeks, the state will narrow the number of organizations administering the vaccine, with hospitals, health systems, pharmacies and FQHCs prioritized. Distribution decisions will also be influenced by each provider’s effectiveness in getting vaccine in eligible 1A arms within seven days of receipt and analysis of COVID-19 impact in the region. Right now, however, the Pennsylvania Department of Health (DOH) is focusing on a course correction to address demand for second dose supply that exceeds total vaccine supply received by the state. Vaccine supply and distribution will be bumpy for a few weeks, but we all hope that predictability and stability of supply will improve thereafter. Sometimes, when the present is challenging and the future hard to envision, it is good to look back. Watch this 3-minute video on polio vaccine rollout.
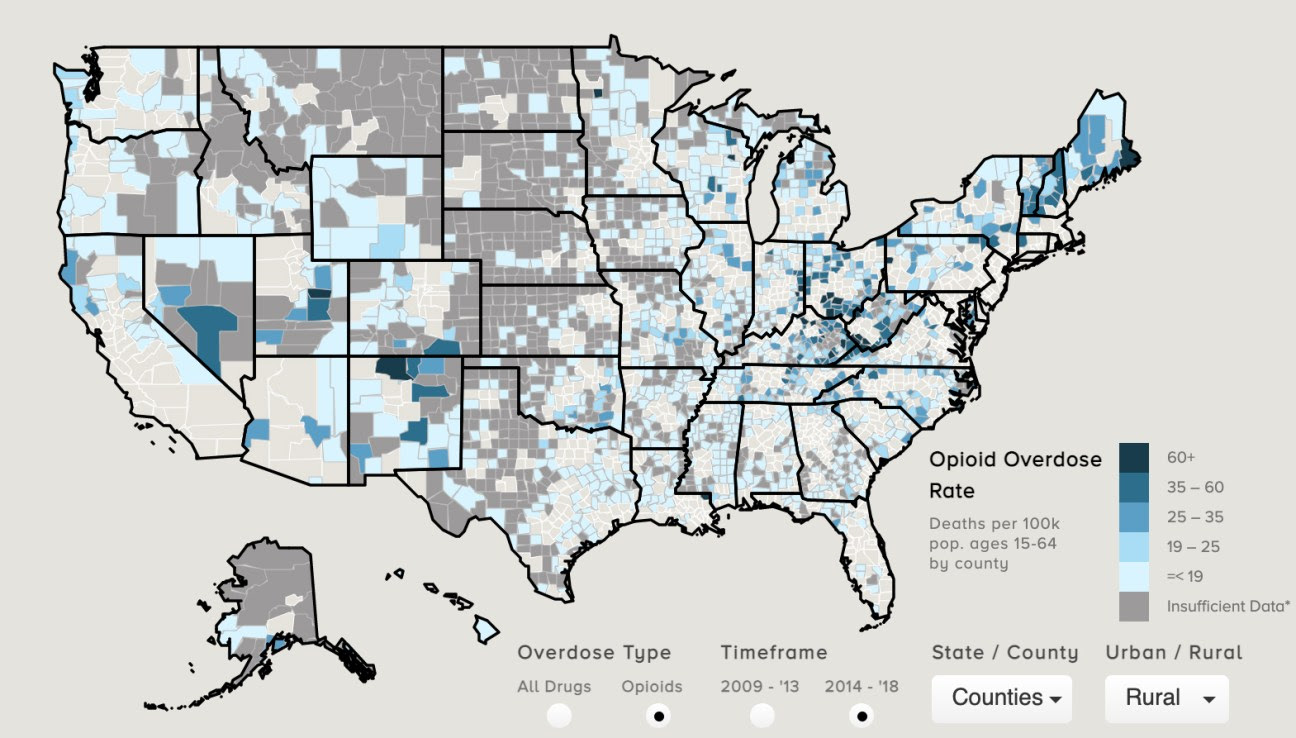
Report: 10 Western States See Some Drug Overdoses Deaths Increase by Nearly 100%

By Liz Carey
The Covid-19 pandemic-related stress, isolation, and a wave of drugs contaminated with extremely potent synthetic opioid, fentanyl, are factors in a severe rise of deadly overdoses.
Read more
Medicaid Policy Trends in 2020: COVID, Coverage and Costs
A recent post from NCSL highlights Medicaid 2020 legislative actions across the country from coronavirus to cost controls―trends that are expected to continue into 2021 sessions.
The NCSL blog post was produced through HRSA’s cooperative agreement with the National Organizations of State and Local Officials (NOSLO).
Improving Access to Care: Medicaid, Telehealth and Health Workforce 101
A post on the NCSL Blog discusses the policy areas of Medicaid, telehealth and health care workforce capacity; and includes relevant state examples and select federal resources for state policymakers aiming to enhance access to health care.
The NCSL blog post was produced through HRSA’s cooperative agreement with the National Organizations of State and Local Officials (NOSLO).
New Research on the Intersection of Pregnancy-Associated Deaths and Intimate Partner Violence
New research examines the three leading causes of pregnancy-associated deaths, describes evidence-based approaches and strategies for prevention and intervention, and makes recommendations for further research.
The research is published in the February 2021 Journal of Women’s Health Special Issue: Maternal Morbidity and Mortality.
The special issue provide a comprehensive review of relevant literature, including biological and physiological risk factors, external risk factors, social determinants of health, and proven and potential interventions.
Sabrina Matoff-Stepp, Ph.D., in HRSA’s Office of Planning, Analysis, and Evaluation and colleagues from NIH, Johns Hopkins University, and the University of Virginia collaborated on the research.
Read the article published in the Special Issue.
Cost-Effectiveness of the Ryan White HIV/AIDS Program
This month, HRSA published a pair of manuscripts demonstrating the cost-effectiveness of the Ryan White HIV/AIDS Program (RWHAP) in the Journal of Acquired Immune Deficiency Syndromes (JAIDS).
These two papers present findings from the first formal evaluation of the RWHAP’s cost-effectiveness at a national level. The first paper presents a new and innovative mathematical model that can be used as a tool for estimating the cost-effectiveness of the RWHAP under a variety of policy scenarios and assumptions. The second paper compares estimated health care costs and outcomes over a 50-year period in the presence of the RWHAP relative to those expected if the comprehensive and integrated system of medical and support services funded by the RWHAP were not available.
Read the first paper.
Read the second paper.
Rural Health System Part of Operation Warp Speed Trials for Covid-19 Treatments

By Liz Carey
On the Canadian border of Upstate New York, a small health system is a rare example of rural-based clinical trials for treating Covid-19.
Read more
Report: To Fix Rural Problems We Have to Overhaul Existing Rural Programs

By Anya Slepyan
Researchers found that the federal development programs aimed at partnering with rural communities are ineffective in the rural environment of today.
Read more
